| TUD Organische Chemie | Immel | Tutorials | Orbitals | Hybrid | Orbitals 2sp2 | View or Print (this frame only) |
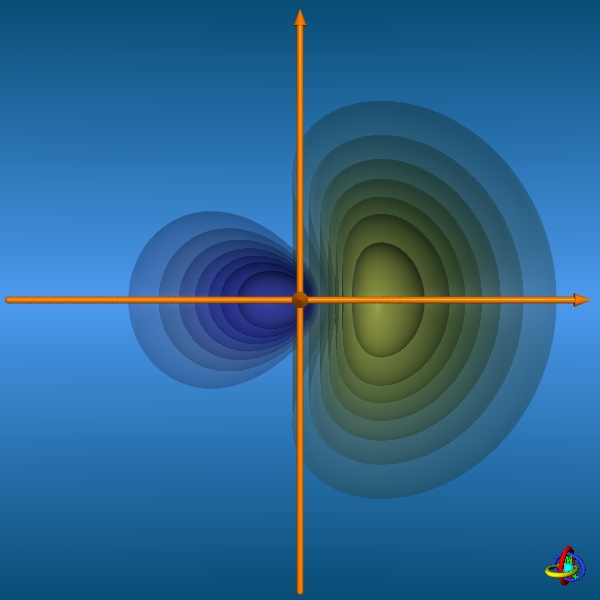
The linear combination of a single 2s-orbital with two 2p-orbitals (here 2px and 2py) results in three 2sp2-orbitals
of identical shape, but different orientation in space. The images below provide an illustration of the probability contours
(90-25% probability level of the electron density, i.e. ψ2) and the nodal plane of a 2sp2-orbital
(actually not a "planar" plane; ψ = 0), where different colors indicate regions of opposite sign in ψ.
The nucleus is located at the origin of the coordinate system as indicated by the orange axes, and thus is embedded not between the lobes
on the nodal plane, but within the minor (blue) lobe of the hybrid orbital.
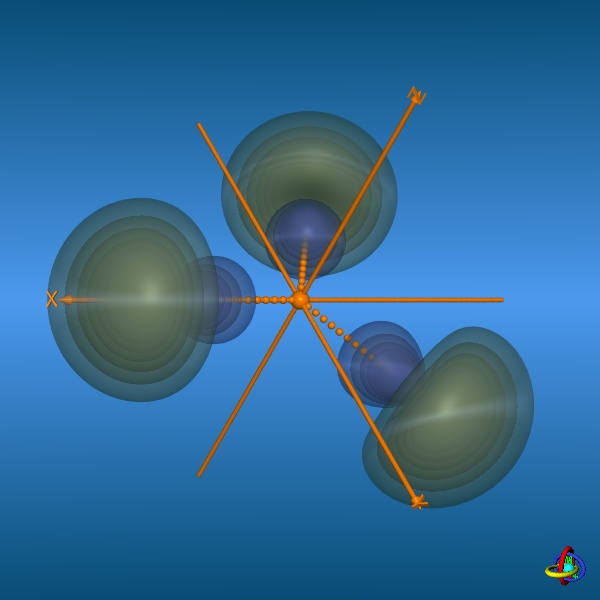
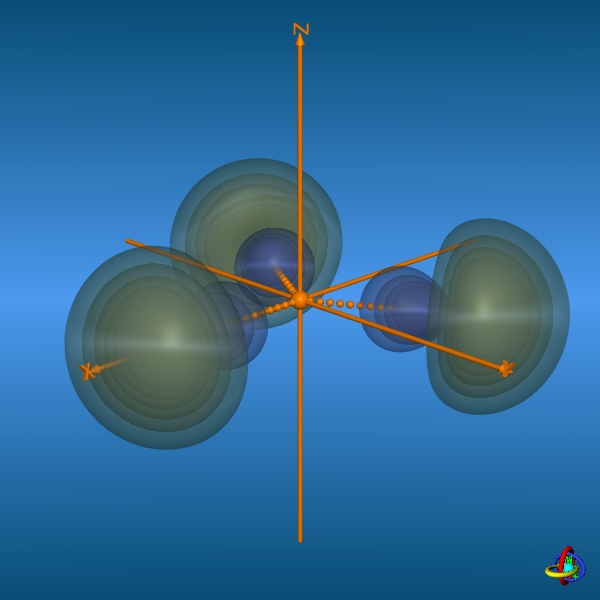
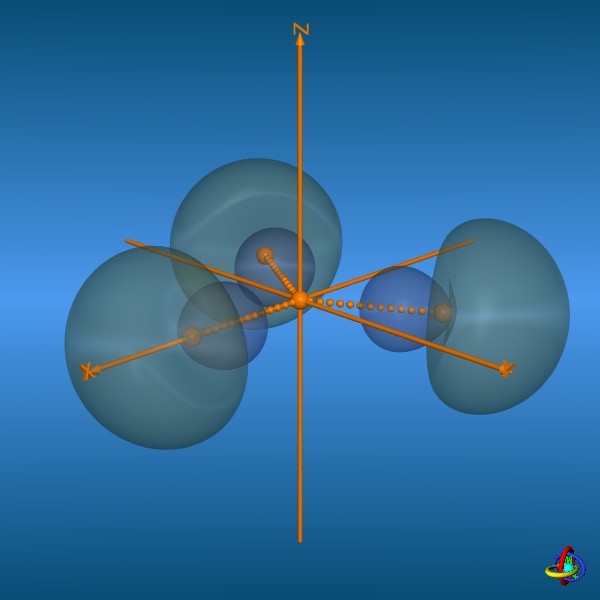
In the bottom row, all three 2sp2-orbitals and their trigonal-planar relative arrangement are displayed, but note that the orbitals have been moved away from the center towards the outside to facilitate visualization (the orange spheres represent the original location of the nucleus). This arrangement of orbitals is suitable to describe the geometry of molecules such as BH3 or alkenes (planarity of H2C=C-fragments with bond angles of 120°). Click on the images to enlarge view or see the VRML 3D-model. |
|||
All graphics shown on this page were created using the MolArch+ program
and POVRAY Persistence of Vision Raytracer. Hybrid Orbitals were generated from the
pure hydrogenic wave functions of 2s- and 2p-orbitals; Cartesian wave functions were taken from
The Orbitron Gallery of Atomic Orbitals and Molecular Orbitals.
|
|||
|
|
|||